Nitrogen Cycle Class 8
Nitrogen cycle is a very important topic, not only from the class 8 perspective but also from the competitive exams point of view.
Importance of Nitrogen
- Nitrogen is the most abundant element found in the earth’s atmosphere i.e. 78 percent.
- Nitrogen cannot be absorbed directly by the plants and animals until it is converted into compounds they can use.
- Plants take Nitrogen in the form of Nitrate.
- Nitrogen is useful in opening and closing of Stomata.
- Deficiency of Nitrogen in plants causes Chlorosis.
What is Nitrogen Cycle?
Nitrogen Cycle Class 8 Definition
A biogeochemical cycle in which Nitrogen and its compounds are interconverted from one form to another as it circulates in the environment.
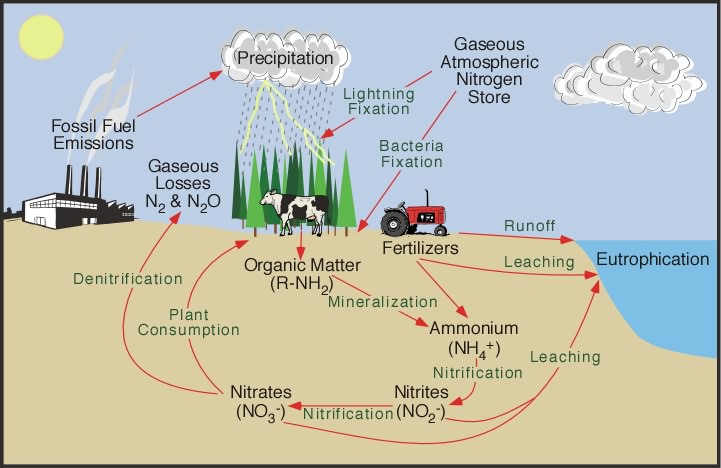
Nitrogen Cycle Steps
It basically involves these following four important processes:-
- Nitrogen Fixation
- Ammonification
- Nitrification
- De-nitrification
Nitrogen Fixation
What is Nitrogen Fixation?
Let us explain the term nitrogen fixation, one of the important processes of Nitrogen Cycle.
The nitrogen molecule (N2) is quite inert. To break it apart so that its atoms can combine with other atoms requires the input of substantial amounts of energy.
The reduction of atmospheric nitrogen (N2) to the ammonium ion (NH+4) is called nitrogen fixation.
Special bacteria convert the nitrogen gas (N2) to ammonia (NH3) which the plants can use.
Three processes are responsible for most of the nitrogen fixation in the biosphere –
- Atmospheric fixation- by lightning
- Industrial fixation
- Biological fixation by certain microbes – Alone or in a symbiotic relationship with some plants and animals.
Ammonification
After all of the living organisms have used the nitrogen, decomposer bacteria convert the nitrogen-rich waste compounds into simpler ones.
It is the formation of Ammonia from Protein and Nucleic Acid.
Bacteria that help- Clostridia, Actinomycetes.
Nitrification
Nitrification is the process which converts the ammonia into nitrite ions which the plants can take in as nutrients.
In short Ammonia to Nitrite ions.
Nitrite to Nitrate (Plant absorbs this nitrate)
De-nitrification
De-nitrification is the final step in which other bacteria convert the simple nitrogen compounds back into nitrogen gas (N2), which is then released back into the atmosphere to begin the cycle again.
In short, Nitrate (the Nitrate which is not absorbed by plants) to Free Nitrogen (N2).
Nitrogen Cycle Diagram
Here is a simple Nitrogen Cycle Diagram.